ThermodynamicsHard
Question
The direct conversion of A to B is difficult, hence it is carried out by the following shown path:
Given ᐃS(A → C) = 50 e.u.
ᐃS(C → D) = 30 e.u.
ᐃS(B → D) = 20 e.u.
where e.u. is entropy unit then ᐃS(A → B) is
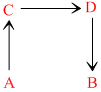
Given ᐃS(A → C) = 50 e.u.
ᐃS(C → D) = 30 e.u.
ᐃS(B → D) = 20 e.u.
where e.u. is entropy unit then ᐃS(A → B) is

Options
A.+ 100 e.u.
B.+ 60 e.u.
C.- 100 e.u.
D.- 60 e.u.
Solution
ᐃS(A → B) = ᐃS(A → C) + ᐃS(C → D) - ᐃS(B → D) = 50 + 30 - 20
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A quantity of 1.6 g helium gas is expanded adiabatically 3.0 times and then compressed isobarically to the initial volum...Each of the vessels 1 and 2 contain 1.2 moles of gaseous helium. The ratio of the volume of vessels is V2/V1 = 2.0, and ...An adiabatic cylinder fitted with an adiabatic piston at the right end of cylinder is divided into two equal halves with...Standard enthalpy of vapourisation ᐃvapH- for water at 100oC is 40.66 kJmol-1. The internal energy ofvapourisation...With what minimum pressure must a given volume of an ideal gas ($\gamma$ = 1.4), originally at 400 K and 100 kPa, be adi...