ThermodynamicsHard
Question
Standard enthalpy of vapourisation ᐃvapH- for water at 100oC is 40.66 kJmol-1. The internal energy ofvapourisation of water at 100oC (in kJ mol-1) is
Options
A.+40.66
B.+37.56
C.-43.76
D.+43.76
Solution
H2O(l) → H2O(g)
V1 of liq. H2O = 18 ml
and V2 of H2O (vap) =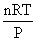
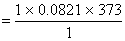 = 30.6233 lit
= 30.6233 lit
ᐃV = (30623.3 ml - 18 ml)
= 30605.3 ml = 30.605 lit
ᐃE = ᐃH - PᐃV
= 40.66 × 103J - 1 × 30.605 × 101.325 J
= 36557.9 = 37.5589 kJ mol-1
V1 of liq. H2O = 18 ml
and V2 of H2O (vap) =
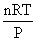
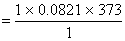 = 30.6233 lit
= 30.6233 litᐃV = (30623.3 ml - 18 ml)
= 30605.3 ml = 30.605 lit
ᐃE = ᐃH - PᐃV
= 40.66 × 103J - 1 × 30.605 × 101.325 J
= 36557.9 = 37.5589 kJ mol-1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The values of ΔG are very important in metallurgy. The ΔG values for the following reactions at 1000 K are given as foll...Which of the following statement(s) is/are correct regarding reversible adiabatic and isothermal processes for an ideal ...A sample of ideal gas is compressed from initial volume of 2Vo to Vo using three different processes as follows. (1) Rev...The minimum work which must be done to compress 16 g of oxygen isothermally at 300 K from a pressure of 1.01325 × 103 N/...Two moles of an ideal gas ($\gamma$ = 1.4) was allowed to expand reversibly and adiabatically from 1 L, 527°C to 32 L. T...