Coordination CompoundHard
Question
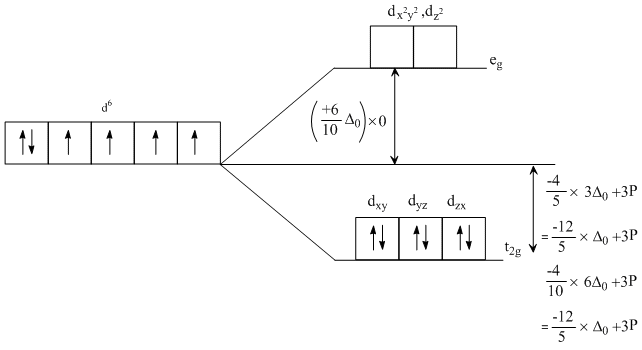
Low spin complex of d6 - cation in an octahedral field will have the following energy:
(ᐃ0 = Crystal Field Splitting Energy in an octahedral field, P = Electron pairing energy)
(ᐃ0 = Crystal Field Splitting Energy in an octahedral field, P = Electron pairing energy)
Options
A.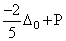
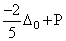
B.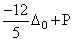
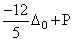
C.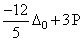
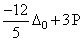
D.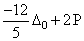
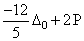
More Coordination Compound Questions
Which of the following is considered to be an anticancer species?...Ni(CO)4 and [Ni(NH3)4]2+ do not differ in...Which of the following are π-bonded organometallic compounds ?...The number of unpaired electrons calculated in [Co(NH3)6]3+ and [CoF6]3– are :...Identify reactions give Diastereo isomeric product ?...