Atomic StructureHard
Question
The percentage of p-character in the orbitals forming P-P bonds in P4 is
Options
A.25
B.33
C.50
D.75
Solution
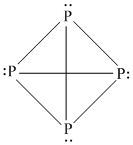
P is sp3 hybridized in P4.
Hence (D) is correct.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A sodium street light gives off yellow light that has a wavelength of 600 nm. Then (For energy of a photon take E = )...The sum of the number of neutrons and proton in the isotope(T) of hydrogen is :...Imagine an atom made up of a stationary proton and a hypothetical particle of double the mass of electron but having the...Radiations of frequency, (ν) are incident on a photosensitive metal. The maximum kinetic energy of photoelectrons is E. ...The radius of La3+ (Atomic number of La = 57) is 1.06 . Which one of the following given values will be closest to the r...