Atomic StructureHard
Question
The radius of La3+ (Atomic number of La = 57) is 1.06 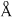 . Which one of the following given values will be closest to the radius of Lu3+ (Atomic number of Lu = 71) ?
. Which one of the following given values will be closest to the radius of Lu3+ (Atomic number of Lu = 71) ?
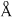 . Which one of the following given values will be closest to the radius of Lu3+ (Atomic number of Lu = 71) ?
. Which one of the following given values will be closest to the radius of Lu3+ (Atomic number of Lu = 71) ?Options
A.1.40 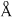
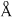
B.1.06 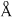
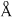
C.0.85 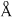
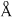
D.1.60 
More Atomic Structure Questions
What should be the increase in kinetic energy of electron in order to decrease its de Broglie wavelength from 100 nm to ...Electromagnetic radiations of wavelength 240 nm are just sufficient to ionize sodium atom. The ionization energy of sodi...A certain dye absorbs light of l = 4000 and then fluorescence light of 5000 . Assuming that under given conditions 50% o...Number of nodal surface in 5s orbital is...An α–particle is accelerated from rest through a potential difference of 6.0V. Its de Broglie wavelength is...
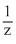
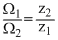 ⇒
⇒ 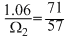 ⇒ Ω2 = 0.85
⇒ Ω2 = 0.85