Chemical BondingHard
Question
The correct stability order for the following species is




Options
A.(II) > (IV) > (I) > (III)
B.(I) > (II) > (III) > (IV)
C.(II) > (I) > (IV) > (III)
D.(I) > (III) > (II) > (IV)
Solution
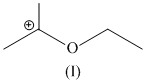
(I)
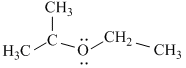
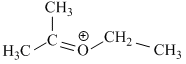
Stabilizes by resonance and have six α - hydrogen atoms (hyperconjugation)
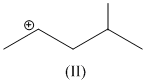
(II)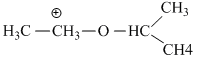
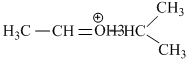
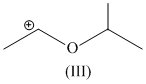
(III) Stabilizes by resonance and have only three α - hydrogen atoms.
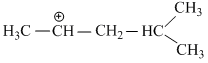
have five α - hydrogen atoms.
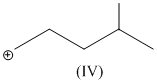
(IV)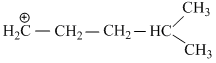
have only two α - hydrogen atoms.
∴ I > III > II > IV
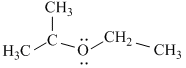
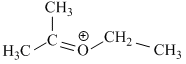
Stabilizes by resonance and have six α - hydrogen atoms (hyperconjugation)
(II)
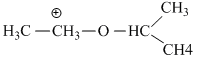
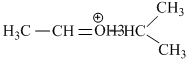
(III) Stabilizes by resonance and have only three α - hydrogen atoms.
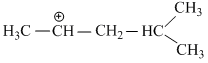
have five α - hydrogen atoms.
(IV)
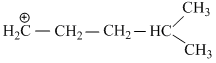
have only two α - hydrogen atoms.
∴ I > III > II > IV
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Among $H_{2}\text{ }S,H_{2}O,{NF}_{3},{NH}_{3}$ and ${CHCl}_{3}$, identify the molecule $(X)$ with lowest dipole moment ...For the following equilibrium H2(g) ⇋ 2H(g)There is 0.1% dissociation at equilibrium pressure of 1 atm. if pressur...Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectivel...In which of the following arrangements the given sequence is not strictly according to the property indicated against it...In XeF2, XeF4, XeF6 the numebr of lone pairs of Xe are respectively...