SolutionHard
Question
The Henry′s law constant for the solubility of N2 gas in water at 298 K is 1.0 × 105 atm. The mole fraction of N2 in air is 0.8. The number of moles of N2 from air dissolved in 10 moles of water at 298 K and 5 atm pressure is
Options
A.4.0 × 10-4
B.4.0 × 10-5
C.5.0 × 10-4
D.4.0 × 10-6
Solution
P = KH χN2
0.8 × 5 = 1 × 105 × χN2
χN2 = 4 × 10-5 (in 10 moles of water)
⇒ 4 × 10-5 =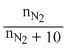
nN2 × 5 × 10-5 + 4 × 10-4 = n N2
⇒ nN2 × = 4 × 10-4
0.8 × 5 = 1 × 105 × χN2
χN2 = 4 × 10-5 (in 10 moles of water)
⇒ 4 × 10-5 =
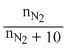
nN2 × 5 × 10-5 + 4 × 10-4 = n N2
⇒ nN2 × = 4 × 10-4
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of pure liquid A is 10 torr and at the same temperature when 1 g of B solid is dissolved in 20 g of ...A quantity of 10 g of solute ‘A’ and 20 g of solute ‘B’ is dissolved in 500 ml water. The solution is isotonic with the ...In a binary electrolyte (AB type), the observed lowering of vapour pressure as compared to the theoretically calculated ...In the following aqueous solutions(A) 1 m sucrose (B) 1 m potassium ferricyanide andC) 1 m potassium sulphate maximum va...Maximum osmotic pressure will be shown by which of the following solution (all 90% ionized)?a) 0.1 M-AlCl3 (b) 0.1 M-BaC...