SolutionHard
Question
The vapour pressure of pure liquid A is 10 torr and at the same temperature when 1 g of B solid is dissolved in 20 g of A, its vapour pressure is reduced to 9.0 torr. If the molecular mass of A is 200 amu, then the molecular mass of B is :
Options
A.100 amu
B.90 amu
C.75 amu
D.120 amu
Solution
R.L.V.P. : 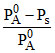 = XB
= XB
XB =
R.L.V.P. :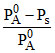 = XB
= XB
XB =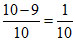
XB =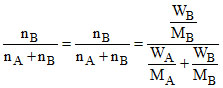
MB =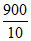 = 90.
= 90.
XB =
R.L.V.P. :
XB =
XB =
MB =
Create a free account to view solution
View Solution FreeMore Solution Questions
The total concentration of dissolved particles inside red blood cells is approximately 0.30 M and the membrane surroundi...Heptane and octane form ideal solution. At 373 K, the vapour pressures of the pure liquids are 106 kPa and 46 kPa, respe...What is molarity of HCl in a solution prepared by dissolving 5.5g HCl in 200 g ethanol. If the density of the solution i...Blood is isotonic with...The molecular mass of glucose is 180 g/ mol and that of sucrose is 342 g/mol. Assuming ideal behaviour, a plot of freezi...