ElectrochemistryHard
Question
The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows:
2/3 Al2O3 → 4/3 Al + O2 ; ᐃrG = + 960kJ mol -1
The potential difference needed for the electrolytic reduction of aluminium oxide (Al2O3) at 500oCat least
2/3 Al2O3 → 4/3 Al + O2 ; ᐃrG = + 960kJ mol -1
The potential difference needed for the electrolytic reduction of aluminium oxide (Al2O3) at 500oCat least
Options
A.5.0 V
B.4.5 V
C.3.0 V
D.2.5 V
Solution
2/3 Al2O3 → 4/3 Al + O2
Al2O6 → 2Al3+ + 3O2-
ᐃG = - nFE ⇒ 960 × 103 = - 6 × 96500 × E
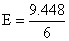 = 1.65 V required
= 1.65 V required
Al2O6 → 2Al3+ + 3O2-
ᐃG = - nFE ⇒ 960 × 103 = - 6 × 96500 × E
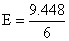 = 1.65 V required
= 1.65 V requiredCreate a free account to view solution
View Solution FreeMore Electrochemistry Questions
At 298K the standard free energy of formation of H2O(l) is -237.20 kJ/mole while ᐃGo of its ionisation into H+ ion...To observer the effect of concentration on the conductivity, electrolytes of different nature are taken in two vessels A...The correct order of EoM2+/M values with negative sign for the four successive elements Cr, Mn, Fe and Co is...During the electrolysis of 0.1 M CuSO4 solution using copper electrodes, a depletion of [Cu++] occurs near the cathode w...Four successive members of the first series of the transition metals are listed below. For which one of them the standar...