Chemical BondingHard
Question
For the reaction equilibrium N2O4 (g) ⇋ 2NO2(g) the concentrations of N2O4 and NO2 at equilibrium are 4.8 × 10-2 and 1.2 × 10-2 mol L-1 respectively. The value of Kc for the reaction is
Options
A.3 × 10-1 mol L-1
B.3 × 10-3 mol L-1
C.3 × 103 mol L-1
D.3.3 × 102 mol L-1
More Chemical Bonding Questions
In the given reaction C6H5 - O - CH2 - CH3 [X] + [Y] [X] and[Y] will respectively be :-...Consider the following sequence of reaction The product B is...Calculate equilibrium concentration ratio of CO(g) and CO2(g). If only CO and H2O are present initially at concentration...The molecules that will have dipole moment are...Which of the following combination(s) lead(s) to the formation of gerade molecular orbital(s) -...
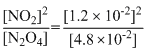 = 3 × 10-3 mol/L
= 3 × 10-3 mol/L