Chemical BondingHard
Question
Calculate equilibrium concentration ratio of CO(g) and CO2(g). If only CO and H2O are present initially at concentration of 0.1 M each?
CO(g) + H2O(g) ⇋ CO2(g) + H2(g) KC = 4.24
CO(g) + H2O(g) ⇋ CO2(g) + H2(g) KC = 4.24
Options
A.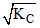
B.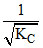
C.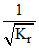
D.2 and 3 both
More Chemical Bonding Questions
A block of mass m moving with a velocity v0 collides with a stationary block of mass M at the back of which a spring of ...Incorrect order according to the property is :...Which of the following best explains the reason for the relative stabilities of the conformers shown?...Which pair have identical bond order :-...The total number of p-bond electrons in the following structure is :-...