Boron and Carbon FamilyHard
Question
What volume of hydrogen gas, at 273 K and 1 atm, pressure will be consumed in obtaining 21.6 g of elemental boron (atomic mass = 10.8) from the reducti on of boron trichloride by hydrogen?
Options
A.67.2 L
B.44.8 L
C.22.4 L
D.89.6 L
Solution
No. of moles of boron = 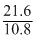 = 2
= 2
for BCl3
∴ 1 mole of Boron = 3 mole of Cl
∴ 2 mole of Boron = 6 mole of Cl
H2 + Cl2 → 2HC
⇒ 3 moles of Hydrogen is required
= 3 × 22.4 = 67.2 Litre
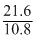 = 2
= 2for BCl3
∴ 1 mole of Boron = 3 mole of Cl
∴ 2 mole of Boron = 6 mole of Cl
H2 + Cl2 → 2HC
⇒ 3 moles of Hydrogen is required
= 3 × 22.4 = 67.2 Litre
Create a free account to view solution
View Solution FreeMore Boron and Carbon Family Questions
For making good quality mirrors, plates of float glass are used. These are obtained by floating molten glass over a liqu...Boric acid...250 m l of a sodium carbonate solution contains2.65 gm. of Na2CO3. If 10 m l of this solution is diluted to one litre. W...A dipeptide is obtained by Glycine & Alanine.wherever it is treated with in presence of sodium carbonate, it gives follo...Number of atoms in 558.5 gram Fe (at.wt. of Fe = 55.85 g mol-1) is...