Boron and Carbon FamilyHard
Question
250 m l of a sodium carbonate solution contains2.65 gm. of Na2CO3. If 10 m l of this solution is diluted to one litre. What is the concentration of the resultant solution?
Options
A.0.1 M
B.0.001 M
C.0.01 M
D.10-4M
Solution
Molarity of solution M1 = 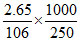 = 0.1 M
= 0.1 M
M1V1 = M2V2
0.1 × 10 = M2 × 1000
M2 =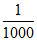 = .001 M
= .001 M
M1V1 = M2V2
0.1 × 10 = M2 × 1000
M2 =
Create a free account to view solution
View Solution FreeMore Boron and Carbon Family Questions
Carbon reduction is not used for extraction of Al from Al2O3 because...Iron carbonyl, Fe(CO)5 is...Which of the following reaction(s) will have final product as hydrocarbon -...The structure of diborane (B2H6) contains...Calculate the partial pressure of carbon monoxide from the following :-CaCO3(s) ⇋ CaO(s) + CO2 (g) ; KP = 8 ×...