Boron and Carbon FamilyHard
Question
Number of atoms in 558.5 gram Fe (at.wt. of Fe = 55.85 g mol-1) is
Options
A.twice that in 60 g carbon
B.6.023 × 1022
C.half that in 8g He
D.558.5 × 6.023 × 1023
Solution
Fe (no. of moles) = 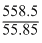 = 10 moles
= 10 moles
C(no. of moles) = 60/12 = 5 moles.
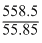 = 10 moles
= 10 molesC(no. of moles) = 60/12 = 5 moles.
Create a free account to view solution
View Solution Free