Solid StateHard
Question
In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the formula of the compound would be if one of the X atoms is missing from a corner in each unit cell
Options
A.X7Y24
B.X24Y7
C.XY24
D.X24Y
Solution
No. of X atom per unit cell = 7 × 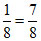
No. of Y atom per unit cell = 6 ×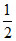 = 3
= 3
No. of X atom per unit cell = 7 ×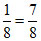
No. of Y atom per unit cell = 6 ×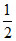 = 3
= 3
∴ Formula = X7/8 Y3 or X7 Y24.
No. of Y atom per unit cell = 6 ×
No. of X atom per unit cell = 7 ×
No. of Y atom per unit cell = 6 ×
∴ Formula = X7/8 Y3 or X7 Y24.
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following oxide shows electrical properties like metals?...Number of 3rd nearest neighbour atom in a FCC unit cell are -...In Na2O structure,...In a compound atoms of element Y from ccp lattice and those of element X occupy 2/3rd of tetrahedral voids. The formula ...Give the correct order of initials T (true) or F (false) for the following statements.I. In an anti-fluorite structure, ...