Solid StateHard
Question
Platinum crystallises in a face centered cube crystal with a unit cell length of 3.9231 A0. The density and atomic radius of platinum are respectively. [Atomic mass of Pt = 195]
Options
A.45.25 g-cm-3, 2.516 A0
B.21.86 g-cm-3, 1.387 A0
C.29.46 g-cm-3, 1.48 A0
D.None of these
Solution
Density = 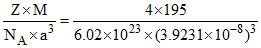 = 21.86 g/cm3
= 21.86 g/cm3
for fcc lattice, 4r = a√2
so, r =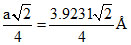 = 1.387 A0
= 1.387 A0
for fcc lattice, 4r = a√2
so, r =
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following are the correct axial distance and axial angles for rhombohedral system?...In a face centred cubic lattice, atom A occupies the corner positions and atom B occupies the face centre positions. If ...In an ionic solid AB2O4, the oxide ions form CCP. ‘A’ and ‘B’ are metal ions in which one is bivalent and another is tri...Edge length of M+X- (fcc structure) is 7.2 A0. Assuming M+ - X- contact along the cell edge, radius of X- ion is(rM+ = 1...The correct statement(s) regarding defects in solids is(are)...