ElectrochemistryHard
Question
What will be the emf for the given cell Pt | H2 (P1) |H+ (aq) | | H2 (P2)| Pt
Options
A.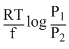
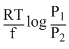
B.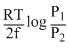
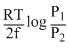
C.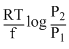
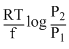
D.none of these
More Electrochemistry Questions
Limiting molar conductivity of NH4OH (i.e., Λom(NH4OH)) is equal to...Electrolysis of a solution of MnSO4 in aqueous sulphuric acid is a method for the preparation of MnO2 as per the reactio...In the balanced chemical reaction, IO3- + aI- + bH+ → cH2O + dI2 a, b, c and d respectively to...The preparation of LiOH by the electrolysis of a 35% solution of LiCl using a platinum anode led to a current efficiency...Consider the cell potentials EoMg2+|Mg = - 2.37 V and EoFe3+|Fe = - 0.04 V. The best reducing agent would be...