Chemical Kinetics and Nuclear ChemistryHard
Question
for a reaction A → B, Ea = 10 kJ mol-1, ᐃH = 5 kJ mol-1. Thus, potential energy profile for this reaction is :
Options
A.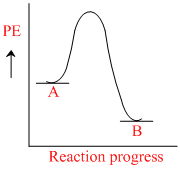

B.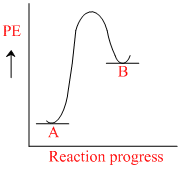

C.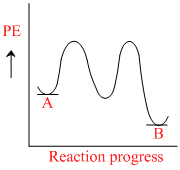

D.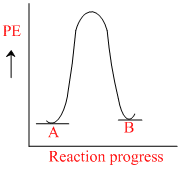

Solution
For reaction, A → B.
Ea = 10 kJ/mole , ᐃH = 5 kJ/mole
Rxn endothermic because ᐃH (+)
ᐃH = Ea - Eb
5 = 10 - Eb
Eab = 10 - 5 = 5 kJ/mole. Then [B].
Ea = 10 kJ/mole , ᐃH = 5 kJ/mole
Rxn endothermic because ᐃH (+)
ᐃH = Ea - Eb
5 = 10 - Eb
Eab = 10 - 5 = 5 kJ/mole. Then [B].
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Consider following reaction, Zn + 2H+ → Zn2+ + H2Half-life period is independent of concentration of Zn at constan...Activation energy of a chemical reaction can be determined by :...Consider the following first-order decomposition reaction. A4(g)→ 4A(g)Which of the following statement(s) is/are correc...The rate law for a reaction between the substances A and B is given by rate = K [A]n [B]m. On doubling the concentration...Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrheni...