Atomic StructureHard
Question
In a hydrogen atom, if energy of an electron in ground state is 13.6 eV, then that in the 2nd excited state is
Options
A.1.51 eV
B.3.4 eV
C.6.04 eV
D.13.6 eV
Solution
2nd excited state will be the 3rd energy level
En =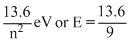 ev = 1.51 eV
ev = 1.51 eV
En =
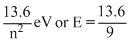 ev = 1.51 eV
ev = 1.51 eVCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
Bohr′s theory is not application to :...Radiation corresponding to the transition n = 4 to n = 2 in hydrogen atoms falls on a certain metal (work function = 2.0...The value of Planck′s constant is 6.63 × 10-34 J-s.The speed of light is 3 × 1017 nm s-1. Which value is closest t...If wavelength is equal to the distance travelled by the electron in one second, then -...Rydberg given the equation for all visible radiation in the hydrogen spectrum as $\lambda = \frac{kn^{2}}{n^{2} - 4}$. T...