Atomic StructureHard
Question
Radiation corresponding to the transition n = 4 to n = 2 in hydrogen atoms falls on a certain metal (work function = 2.0 eV). The maximum kinetic energy of the photoelectrons will be :-
Options
A.0.55 eV
B.2.55 eV
C.4.45 eV
D.None
Solution
En = - 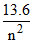 eV;E2 =
eV;E2 = 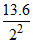 ;E4 =
;E4 = 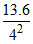 eV/atom
eV/atom
ᐃE = E4 - E2 = 2.55 eV
Absorbed energy = work function of metal + K.E.
2.55 = 2 + K.E.
K.e. = 0.55 eV
ᐃE = E4 - E2 = 2.55 eV
Absorbed energy = work function of metal + K.E.
2.55 = 2 + K.E.
K.e. = 0.55 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The energy of an electron in the first Bohr's orbit of H-atom is –13.6 eV. The possible energy value(s) of the excited s...The quantum numbers +1/2 and –1/2 for the electron spin represent...If the mass of electron is doubled, the speed of electron revolving around Li2+ nucleus will...Match the following(a) Energy of ground state of He+ (i) + 6.04 eV(b) Potential energy of I orbit of H-atom (ii) -27.2 e...How many of the following compounds have used their d-orbital in hybridisationCF4, SF4, I3-, SOF4, PF3Cl2, SnCl2, SO42-...