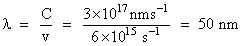Atomic StructureHard
Question
The value of Planck′s constant is 6.63 × 10-34 J-s.The speed of light is 3 × 1017 nm s-1. Which value is closest to the wavelength in nanometer of aquantum of light with frequency of 6 × 1015 s-1 ?
Options
A.75
B.10
C.25
D.50
More Atomic Structure Questions
How many unpaired electrons are present in the ground state of chromium (Z = 24)?...The radius of first orbit of H-atoms is 0.529 Å. The radius of first orbit of D-atoms should be...The total number of orbitals in a shell with principal quantum number ′n′ is :...Mny elements have non-integral atomic masses because :...If element 25X+Y has spin magnetic moment 1.732 B.M then...