Atomic StructureHard
Question
In a certain electronic transition in the hydrogen atoms from an initial state (1) to a final state (2), the difference in the orbital radius (r1 - r2) is 24 times the first Bohr radius. Identify the transition.
Options
A.5 → 1
B.25 → 1
C.8 → 3
D.6 → 5
Solution
r1 - r2 = 24 × (r1)H
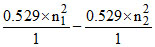 = 24 × 0.529
= 24 × 0.529
∴ (n12 - n22) = 24
So, n1 = 5 and n2 = 1
∴ (n12 - n22) = 24
So, n1 = 5 and n2 = 1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Given that abundances of isotopes 54Fe, 56Fe, and 57Fe are 5%, 90% and 5% respectively, the atomic mass of Fe is :-...The number of nodal planes in a Px orbotal is :...In [Cu(NH3)4]2+ ion the unpaired electron is present is :-...If the shortest wave length of Lyman series of H atom is x, then the wave length of the first line of Balmer series of H...The group in which all the elements do not have same number of electrons in their valence shell is :-...