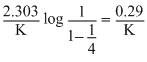Chemical Kinetics and Nuclear ChemistryHard
Question
t1/4 can be taken as the time taken for the concentration of a reactant to drop to 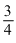 of its initial value. If the rate constant for a first order reaction is K, the t1/4 can be written as
of its initial value. If the rate constant for a first order reaction is K, the t1/4 can be written as
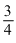 of its initial value. If the rate constant for a first order reaction is K, the t1/4 can be written as
of its initial value. If the rate constant for a first order reaction is K, the t1/4 can be written asOptions
A.0.10 / K
B.0.29 / K
C.0.69 / K
D.0.75 / K
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the elementary reaction M → N, the rate of disappearance of M increases by a factor of 8 upon doubling the con...Purple of cassius is colloidal solution of :...Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure. The corr...The following data certain to reaction between A and B ; Which of the following inference(s) can be drawn from the above...The formation of gas at the surface of tungsten due to adsorption is the reaction of order...