Coordination CompoundHard
Question
What is the shape of Fe(CO)5 molecule ? Given that its dipole moment = 0.
Options
A.Tetrahedral
B.Octahedral
C.Trigonal bipyramidal
D.Square pyramidal
Solution
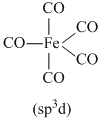 Trigonalbipyramidal ; being symmetrical molecule μ = 0.
Trigonalbipyramidal ; being symmetrical molecule μ = 0.As it is symmetrical molecule, therefore, its dipole moment (μ) is zero.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
In which of the following pairs of complexes the central metals/ions do have same effective atomic number ?...Which of the following complexes is not a chelate ?...All the metal ions contains t2g 6eg0 configurations. Which of the following complex will be paramagnetic?...CuSO4 and KCN react to produce :...The equation which is balanced and represents the correct product(s) is...