Coordination CompoundHard
Question
Which of the following complex is not correctly matched with its geometry ?
Options
A.[NiCl2(Ph3P)2] - tetrahedral
B.[Co(Py)4]2+ - square planar.
C.[Cu(CN)4]3- - tetrahedral
D.[Fe(CO)4]2- - square planar.
Solution
In complex the iron is in -2 oxidation state. CO is strong field ligand and, therefore pairing of electrons occurs. So,
[Fe(CO)4]2 :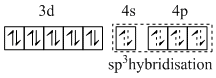
[Fe(CO)4]2 :
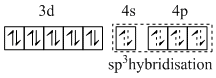
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following complexes produces three moles of silver chloride when its one mole is treated with excess of sil...Which one of the following complexes exhibit chirality ?...Which of the following complex has zero magnetic moment (spin only)?...Silver cholride dissolves in :...Philosopher′s wool on heating with BaO at 1100oC produces:...