SolutionHard
Question
The solubility of Al(OH)3 and Zn(OH)2 are 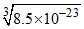 and 1.8 × 10-14, respectively. If NH4OH is added to a solution containing Al3+ and Zn2+ ions, then substance precipitated first is
and 1.8 × 10-14, respectively. If NH4OH is added to a solution containing Al3+ and Zn2+ ions, then substance precipitated first is
Options
A.Al(OH)3
B.Zn(OH)2
C.Both (a) and (b)
D.None of these
Solution
(Assume [Al3+] = [Zn2+])
The compound which have less Ksp value will be precipitated first. Since the Ksp of Al(OH)3 is less than Ksp of Zn(OH)2, So former is precipitated.
[OH-]min for Al3+ =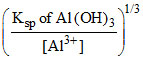
[OH-]min for Al2+ =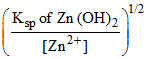
The compound which have less Ksp value will be precipitated first. Since the Ksp of Al(OH)3 is less than Ksp of Zn(OH)2, So former is precipitated.
[OH-]min for Al3+ =
[OH-]min for Al2+ =
Create a free account to view solution
View Solution FreeMore Solution Questions
An aqueous solution of gas (X) gives the white turbidity on passing H2S in the solution. Identify (X)...Consider the following aqueous solutions.I. 2.2 g Glucose in 125 mL of solution.II. 1.9 g Calcium chloride in 250 mL of ...The solution having higher osmotic pressure than the reference solution is called...In which of the following pairs of solutions will the values of the vant Hoff factor be the same?...The mass of KCl required to depress the freezing point of 500 g water by 2 K is (Kf = 1.86, K = 39)...