Mole ConceptHardBloom L3
Question
The density (in g mL-1) of a 3.60 M sulphuric acid solution that is 29% H2SO4 (Molar mass = 98 g mol-1) by mass will be
Options
A.1.64
B.1.88
C.1.22
D.1.45
Solution
Let the density of solution be ′d′
Molarity of solution given = 3.6
i.e. 1 litre of solution contains 3.6 moles of H2SO4
or 1 litre of solution contains 3.6 × 98 gms of H2SO4
Since, the solution is 29% by mass.
100 gm solution contains 29 gm H2SO4
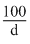 mi solution contains 29 gm of H2SO4
mi solution contains 29 gm of H2SO4
1000 ml solution contains 3.6 × 98 gm of H2SO4
∴ 3.6 × 98 =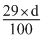 × 1000
× 1000
d = 1.22
Hence, (3) is correct.
Molarity of solution given = 3.6
i.e. 1 litre of solution contains 3.6 moles of H2SO4
or 1 litre of solution contains 3.6 × 98 gms of H2SO4
Since, the solution is 29% by mass.
100 gm solution contains 29 gm H2SO4
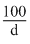 mi solution contains 29 gm of H2SO4
mi solution contains 29 gm of H2SO41000 ml solution contains 3.6 × 98 gm of H2SO4
∴ 3.6 × 98 =
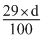 × 1000
× 1000 d = 1.22
Hence, (3) is correct.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
16 g of an ideal gas SOx occupies 5.6 L. at STP. The value of x is...For preparing 0.1 N solution of a compound from its impure sample sample of which the percentage purity is known, the we...V1 ml of unknown gas (A) + V2 ml of O2 → (V1 + V2) ml of CO2. Gas ‘A’ may be...Chlorofluorocarbons such as CCl3F (M = 137.5) and CCl2F2 (M = 121) have been linked to ozone depletion in Antarctica. As...Silver metal in ore is dissolved by potassium cyanide solution in the presence of air by the reaction 4 Ag + 8 KCN + O2 ...