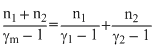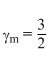ThermodynamicsHard
Question
One mole of ideal monoatomic gas (γ = 5/3) is mixed with one mole of diatomic gas(γ = 7/5). What is γ for the mixture ? γ denotes the ratio of specific heat at constant pressure, to that at constant volume.
Options
A.3/2
B.23/15
C.35/23
D.4/3
More Thermodynamics Questions
The volume of an ideal gas increases 8 times and temperature becomes $(1/4)^{\text{th~}}$ of initial temperature during ...Most accurate values are :-...70 cal of heat are required to raise the temperature of 2 moles of an ideal diatomic gas at constant pressure 30oC to 35...Which of the following graph correctly represent the variation of with p for an ideal gas at constant temperature?...A gas is taken through the cycle A → B → C → A, as shown, What is the net work done by the gas ?...