SolutionHard
Question
The freezing point (in oC) of a solution containing 0.1 g of K3[ Fe(CN)6 ] (Mol. Wt. 329) in 100 g of water (Kf = 1.86 K kg mol-1) is
Options
A.- 2.3 × 10-2
B.- 5.7 × 10-2
C.- 5.7 × 10-3
D.- 1.2 × 10-2
Solution
K3[Fe(CN)6] → 3K+ + [Fe(CN)6]3-
i = 4
ᐃTf = Kf × i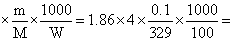 2.3 × 10-2
2.3 × 10-2
T′f = - 2.3 × 10-2
i = 4
ᐃTf = Kf × i
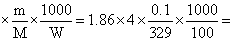 2.3 × 10-2
2.3 × 10-2 T′f = - 2.3 × 10-2
Create a free account to view solution
View Solution FreeMore Solution Questions
The molarity of H2SO4 solution, which has a density 1.84g/cc. at 35oC and contains 98% by weight, is...Which loses weight on exposure to the atmosphere:-...300 ml, N/100 HNO3 solution is mixed with 200 ml, N/100 KOH solution. The [H+] in the resultant solution is :-...Two aqueous solutions S2 and S2 are separated by a semipermeable membrane. S1 has lower vapour pressure than S2. Which o...A liquid mixture of ‘A’ and ‘B’ (assume ideal solution) is placed in a cylinder and piston arrangement. The piston is sl...