ThermodynamicsHard
Question
As shown in the diagram for a closed path ABCA :
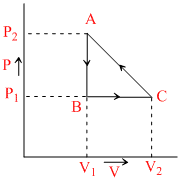

Options
A.The amount of work done by the system is zero
B.The amount of work done by the system is = − 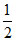 (P2 − P1) (V2 − V1)
(P2 − P1) (V2 − V1)
C.The amount of work done on the system is = (P2 − P1) (V1 − V2)
D.The amount of work done by the system is = 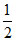 (P2 − P1) (V2 − V1)
(P2 − P1) (V2 − V1)
More Thermodynamics Questions
Specific heat of a gas undergoing adiabatic change is :...In the isothermal expansion of a gas :...In the given graph the isothermal curves are :-...A system does 50 J of work under adiabatic condition. In this process :-...One mole of an ideal atomic gas is heated at a constant pressure of one atmosphere from 0oC to 100oC. Then the change in...