ThermodynamicsHard
Question
Four curves A, B, C and D are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes are :-
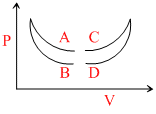

Options
A.C and D respectively
B.D and C respectively
C.A and B respectively
D.B and A respectively
More Thermodynamics Questions
A black body has maximum wavelength λm at 2000 K. Its corresponding wavelength at 3000 K is:...Which of the following parameters does not characterize the thermodynamic state of matter?...An ideal gas is subjected to an isothermal expansion such that its volume change fromVi to Vf and pressure from Pi to Pf...The real coefficient of volume expansion of glycerine is 0.000597 per oC and linear coefficient of expansion of glass is...If 10 gm ice at 0oC is mixed with 10 gm water at 20oC, the final temperature will be :-...