Solid StateHard
Question
In a compound atoms of element Y from ccp lattice and those of element X occupy 2/3rd of tetrahedral voids. The formula of the compound will be
Options
A.X4Y3
B.X2Y3
C.X2Y
D.X3Y4
Solution
No. of atoms of Y = 4
No. of atoms of X =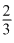 × 8
× 8
Formula of compound will be X4Y3
No. of atoms of X =
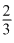 × 8
× 8Formula of compound will be X4Y3
Create a free account to view solution
View Solution FreeMore Solid State Questions
An element (atomic = 100 g/mol) having bcc structure has unit cell edge 400 pm. Density of the element is...A transition metal M can exist in two oxidation states +2 and +3. It forms an oxide whose experimental formula is given ...Cesium chloride on heating to 760 K changes into...Lithium crystallizes in a body centered cubic lattice. How many next–nearest neighbors does each Li have?...In a crystal lattice of BaF2, the C.N of Ba+2 ion is 8. The CO-oxidination number of fluoride (F-) ion must be :-...