Solid StateHard
Question
A transition metal M can exist in two oxidation states +2 and +3. It forms an oxide whose experimental formula is given by MxO where x < 1.Then the ratio of metal ions in +3 state to those in +2 state is given by:
Options
A.(1 - x)/(1 + x)
B.1 + 2x
C.1 + x/2
D.2(1 - x) / (3x - 2)
Solution
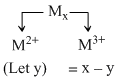
⇒ Applying charge balance , 2y + 3(x - y) - 2 = 0
⇒ y = 3x - 2
∴
Create a free account to view solution
View Solution FreeMore Solid State Questions
If the radii of A+ and B- are 95 pm and 181 pm respectively, then the coordination number of A+ will be ?...A metal crystallizes in such a lattice in which only 70% of the total space of the crystal is occupied by the atoms. If ...Antifluorite structure is derived from fluorite structure by :...Copper crystallizes in fcc with a unit cell length of 361 pm. What is the radius of copper atom?...A mineral having the formula AB2, crystallises in the cubic close - packed lattice, with the A atoms occupying the latti...