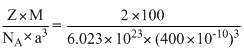Solid StateHard
Question
An element (atomic = 100 g/mol) having bcc structure has unit cell edge 400 pm. Density of the element is
Options
A.10.376 g/ cm3
B.5.188 g/ cm3
C.7.289 g/ cm3
D.2.144 g/ cm3
More Solid State Questions
A binary solid (A+ B−) has a rock salt structure. If the edge length is 400 pm and the radius of cation is 75 pm, then t...NaCl is doped with 2 × 10–3 mole% SrCl2, the concentration of cation vacancies is (NA = 6 × 1023)...The correct information regarding a solid element (atomic radius = r) having ABCABC…. type packing (FCC unit cell edge l...A given metal crystallizes out with a cubic structure having edge length of 361 pm. If there are four metal atoms in one...There are three cubic unit cells A, B and C. A is FCC and all of its tetrahedral voids are also occupied. B is also FCC ...