Chemical EquilibriumHard
Question
The equilibrium constants KP1 and KP2 for the reactions X ⇋ 2Y and Z ⇋ P + Q, respectively are in the ratio of 1 : 9. If the degree of dissociation of X and Z be equal then the ratio of total pressure at these equilibria is
Options
A.1 : 36
B.1 : 1
C.1 : 3
D.1 : 9
Solution
X ⇋ 2Y
1 0
(1 - x) 2x
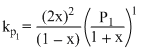
Z ⇋ P + Q
1 0 0
(1 - x) x x
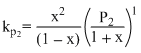
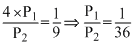
1 0
(1 - x) 2x
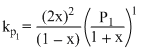
Z ⇋ P + Q
1 0 0
(1 - x) x x
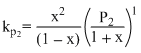
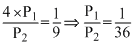
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
One mole of He (g, colourless) is added to the equilibrium mixture containing N2O4 (g, colourless) and NO2 (g, red-brown...Which of the following reaction(s) have KP = KC?...The first and second dissociation constants of an acid H2A are 1.0 × 10-5 and 5.0 × 10-10 respectively.The ove...In a vessel of 1.0 L capacity, O2(g) at 0.25 atm pressure and HCl(g) at 1.0 atm pressure are allowed to react in the pre...X2(g) + Y2(g) ⇋ 2XY(g) reaction was studied at a certain temperature. In the beginning 1 mole of X2 was taken in a...