JEE Advanced | 2018Atomic StructureHard
Question
One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (whre V is the volume and T is the temperature). Which of the statements below is (are) true ?
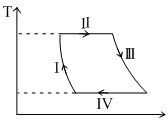
Options
A.
Process I is an isochoric process
B.
In process II, gas absorbs heat
C.
In process IV, gas releases heat
D.
Processes I and II are not isobaric
Solution
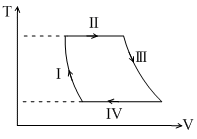
(A) Process-I is not isochoric, V is decreasing.
(B) Process-II is isothermal expansion
U = 0, W > 0
Q > 0
(C) Process-IV is isothermal compression,
U = 0, W < 0
Q < 0
(D) Process-I and III are NOT isobaric because in isobaric process T V hence isobaric T–V graph
will be linear.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The half life of a radioactive substance against α - decay is 1.2 × 107 s. What is the decay rate for 4.0 ...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...The work function of a substance is $4.0$ eV. The longest wavelength of light that can cause photoelectron emission from...In a hydrogen like atom electron makes transition from an energy level with quantum number n to another with quantum num...The X-ray beam coming from an X-ray tube will be...