Redox and Equivalent ConceptHard
Question
A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number ?
Options
A.S
B.H
C.Cl
D.C
Solution
When a mixture of potassium chlorate, oxalic acid and sulphuric acid is heated, the following reaction occurs :
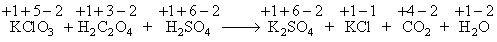
Thus, Cl is the element which undergoes maximum change in the oxidation state.
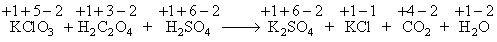
Thus, Cl is the element which undergoes maximum change in the oxidation state.
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
The number of moles of KMnO4 that will be needed to react with one mole of sulphite ion in acidic solution is :...The number of moles of Cr2O72− needed to oxidize 0.136 equivalent of N2H5+ through the reactionN2H5+ + Cr2O72− → N2 + Cr...Equivalent weight of a metal is 18.67. When it reacts with chlorine, the mass of metal which will form 162.52 g of metal...In the reaction, Pb + PbO2 + H2SO4 → PbSO4 + H2O, the equivalent weight of H2SO4 is...Which of the following is not a disproportionation reaction ?...