Redox and Equivalent ConceptHard
Question
Which of the following is not a disproportionation reaction ?
Options
A.2PhCHO 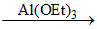 PhCOOCH2Ph
PhCOOCH2Ph
B.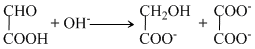
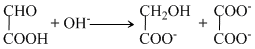
C.NaH + H2O → NaOH + H2
D.All
Solution
(a) and (b) are disproportionation reactions.
(c) is an oxidation reaction.
2H- → H2 + 2e-
(c) is an oxidation reaction.
2H- → H2 + 2e-
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
A mole of N2H4 loses 10 mol of electrons to from a new compound y. Assuming that all the nitrogen appears in the new com...Calculate equivalent weight of KHC2O4 (potassium acid oxalate) when :-(a) It is neutralized by KOH(b) It is oxidised by ...When copper oxide is strongly heated with hydrogen, it reduces to copper. The loss in its weight is 14.9 g and the weigh...What volume of 0.40 M-Na2S2O3 would be required to react with the I2 liberated by adding excess of KI to 50 ml of 0.20 M...The pair of compounds having metals in their highest oxidation state is...