NEET | 2016Nitrogen Containing CompoundHard
Question
The correct statement regarding the basicity of aryl amines is:
Options
A.
Aryl amines are generally less basic than alkyl amines because the nitrogen lone-pair electrons aredelocalized by interaction with the aromatic ring π electron system.
B.
Aryl amines are generally more basic than alkyl amines because the nitrogen lone-pair electrons are notdelocalized by interaction with the aromatic ring π electron system.
C.
Aryl amines are generally more basic than alkyl amines because of aryl group.
D.
Aryl amines are generally more basic than alkyl amines, because the nitrogen atom in aryl amines is sp-hybridized.
Solution
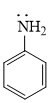
Here lone pair is in conjugation with double bond so basic strength decreased
R - NH2 (No conjugation)
Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
Lowest boiling point will be of the compound-...Considering the basic strength of amines in aqueous solution, which one has the smallest pKb value?...A secondary amine is a compound which possesses-...p-chloroaniline and anilinium hydrochloride can be distinguished by:...Which of the following compound gives the smell of mustard oil-...