JEE Main | 2014Nitrogen Containing CompoundHard
Question
Considering the basic strength of amines in aqueous solution, which one has the smallest pKb value?
Options
A.(CH3)2NH
B.CH3NH2
C.(CH3)3N
D.C6H5NH2
Solution
Among C6H5NH2, CH3NH2, (CH3)2NH,
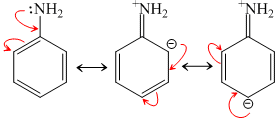
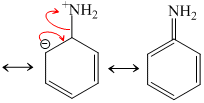
(CH3)3N. C6H5NH2 is least basic due to resonance.
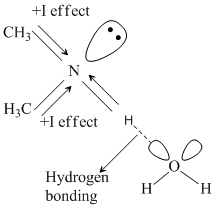
Out of (CH3)3N, CH3NH2, (CH3)2NH. (CH3)2NH is most basic due to +I effect and hydrogen bonding in H2O.
(CH3)3N. C6H5NH2 is least basic due to resonance.


Out of (CH3)3N, CH3NH2, (CH3)2NH. (CH3)2NH is most basic due to +I effect and hydrogen bonding in H2O.

Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
The presence of primary amines can be confirmed by-...The products (A) and (B) formed in the given reaction is RCOCl (A) (B)...An optically active compound (A) decolourises Br2/CCl4 and releases N2 with nitrous acid. The compound (A) is :...Among the following, the strongest base is :...When an organic compound was treated with sodium nitrite and HCl in the ice-cold condition, nitrogen gas was evolved. Th...