JEE Advanced | 2017Atomic StructureHard
Question
Among the following, the correct statement(s) is(are)
Options
A.
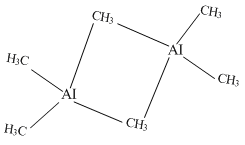
Al(CH3)3 has the three-centre two-electron bonds in its dimeric structure
B.
BH3 has the three-centre two-electron bonds in its dimeric structure
C.
AlCl3 has the three-centre two-electron bonds in its dimeric structure
D.
The Lewis acidity of BCl3 is greater than that of AlCl3
Solution
Both Al(CH3)3 and BH3 has 3c – 2e bonds in the dimeric structure.
BCl3 is stronger Lewis acid than AlCl3.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The ionization enthalpy of hydrogen atom is 1.312 × 106 Jmol-1. The energy required to excite the electron in the a...A hydrogen - like atom has ground state binding energy 122.4 eV. Then :...A + 3B + 3C ⇋ AB2C3Reaction of 6.0 g of A, 6.0 × 1023 atoms of B, and 0.036 mol of C yields 4.8 g of compound...Bond dissociation on energy of Br2 is 200kJ/mole. The longest wavelength of photon that can break this bond would be (NA...Orbital with maximum symmetry is...