Atomic StructureHard
Question
A + 3B + 3C ⇋ AB2C3
Reaction of 6.0 g of A, 6.0 × 1023 atoms of B, and 0.036 mol of C yields 4.8 g of compound AB2C3. If the atomic mass of A and C are 60 and 80 amu, respectively, the atomic mass of B is (Avogadro no. = 6 ×1023):
Reaction of 6.0 g of A, 6.0 × 1023 atoms of B, and 0.036 mol of C yields 4.8 g of compound AB2C3. If the atomic mass of A and C are 60 and 80 amu, respectively, the atomic mass of B is (Avogadro no. = 6 ×1023):
Options
A.50 amu
B.60 amu
C.70 amu
D.40 amu
Solution
nA = 0.1, nB = 1, nC = 0.036
Limiting reagent = C
⇒ nAB2C3 formed =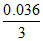 = 0.012
= 0.012
⇒ MM(AB2C3)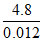 = 400
= 400
⇒ 60 + 2x + 80 × 3 = 400
x = 50
Limiting reagent = C
⇒ nAB2C3 formed =
⇒ MM(AB2C3)
⇒ 60 + 2x + 80 × 3 = 400
x = 50
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The group in which all the elements do not have same number of electrons in their valence shell is :-...O2 undergoes photochemical dissociation into one normal oxygen atom and one oxygen atom, 1.2 eV more energetic than norm...Wrong statement about [Ni(CO)4] is :-...Bohr’s model may be applied to...The correct set of quantum numbers defining the highest energy electron in scandium (I) ion is...