Atomic StructureHard
Question
The ionization enthalpy of hydrogen atom is 1.312 × 106 Jmol-1. The energy required to excite the electron in the atom from n = 1 to n = 2 is
Options
A.8.51 × 105 j mol-1
B.6.56 × 105 j mol-1
C.7.56 × 105 j mol-1
D.9.84 × 105 j mol-1
Solution
ᐃE = E2 - E1 = - 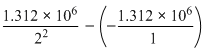
= 9.84 × 105 J mol-1
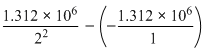
= 9.84 × 105 J mol-1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
What are the number of atoms per unit cell and the number of nearest neighbours in a body centered cubic structure?...Which of the following energy level may bring absorption of photon but never emission of photon?...In which of the following orbital, electron will be closer to the nucleus?...The radionucleide undergoes two successive β - decays followed by one α - decay. The atomic numberand the mass...The most probable radius (in pm) for finding the electron in He+ is...