JEE Main | 2014Mole ConceptHard
Question
The ratio of masses of oxygen and nitrogen in a particular gaseous mixture is 1 : 4. The ratio of number of their molecule is
Options
A.1 : 4
B.7 : 32
C.1 : 8
D.3 : 16
Solution
Let the mass of O2 = x
Mass of N2 = 4x
Number of moles of O2 =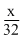
Number of moles of N2 =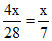
∴ Ratio =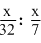 = 7 : 32
= 7 : 32
Mass of N2 = 4x
Number of moles of O2 =
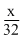
Number of moles of N2 =
∴ Ratio =
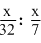 = 7 : 32
= 7 : 32Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The mineral haematite is Fe2O3. Haematite ore contains unwanted material called gangue in addition to Fe2O3. If 5 kg of ...The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60 u) in 1000 g of water is 1.15 g/mL. The m...One mole of ${Cl}_{2}(\text{ }g)$ was passed into 2 L of cold 2 M KOH solution. After the reaction, the concentrations o...Vapour density of a volatile substance is 1.2 (C2H6 = 1). Its molecular mass would be...1.0 g of magnesium is burnt with 0.56 g O2 in a closed vessel. Which reactant is left in excess and how much? (At. wt. M...