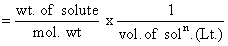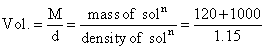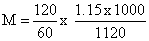Mole ConceptHard
Question
The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60 u) in 1000 g of water is 1.15 g/mL. The molarity of this solution is :-
Options
A.2.05 M
B.0.50 M
C.1.78 M
D.1.02 M
More Mole Concept Questions
SO2 gas is slowly passed through an aqueous suspension containing 12 g of CaSO3 till the milkiness just disappears. What...In which molecule all X - O bond length present in molecule are equal (X = central atom) :-...When a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and aluminium oxide. In an e...10 g of CaCO3 contains :-...For CuSO4·5H2O, which is the correct mole relationship?...