JEE Advanced | 2014ThermodynamicsHard
Question
For complete combustion of ethanol,
C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l),
the amount of heat produced as measured in bomb calorimeter, is 1364.47 kJ mol-1 at 25oC. Assuming ideality the enthalpy of combustion, ᐃcH, for the reaction will be
(R = 8.314 J mol-1)
C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l),
the amount of heat produced as measured in bomb calorimeter, is 1364.47 kJ mol-1 at 25oC. Assuming ideality the enthalpy of combustion, ᐃcH, for the reaction will be
(R = 8.314 J mol-1)
Options
A.-1366.95 kJ mol-1
B.- 1361.95 kJ mol-1
C.- 1460.50 kJ mol-1
D.- 1350.50 kJ mol-1
Solution
C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l)
Bomb calorimeter gives ᐃU of the reaction
So, as per question
ᐃU = - 1364.47 kJ mol-1
ᐃng = - 1
ᐃH = ᐃU + ᐃngRT
= - 1364.47 -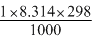
= - 1366.93 kJ mol-1
Bomb calorimeter gives ᐃU of the reaction
So, as per question
ᐃU = - 1364.47 kJ mol-1
ᐃng = - 1
ᐃH = ᐃU + ᐃngRT
= - 1364.47 -
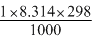
= - 1366.93 kJ mol-1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In any natural process :-...The standard Gibbs energy change at 300 K for the reaction 2A ⇋ B + C is 2494.2J. At a given time, the composition...A system absorbs 100 kJ heat in the process shown in the figure. What is ΔU for the system?...For an isolated system, the wall/boundary separating the system from surrounding must be...A gas is heated at constant pressure. The fraction of heat absorbed used in doing work is...