JEE Main | 2014ElectrochemistryHard
Question
Resistance of 0.2 M solution of an electrolyte is 50 Ω. The specific conductance of the solution is 1.4 S m-1. The resistance of 0.5 M solution of the same electrolyte is 280 Ω. The molar conductivity of 0.5 M solution of the electrolyte in S m2 mol-1 is
Options
A.5 × 10-4
B.5 × 10-3
C.5 × 103
D.5 × 102
Solution
For 0.2 M solution
R = 50 Ω
σ = 1.4 Sm-1 = 1.4 × 10-2 S cm-1]
⇒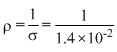 Ω cm
Ω cm
Now, R = ρ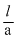
⇒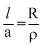 = 50 × 1.4 × 10-2
= 50 × 1.4 × 10-2
For 0.5 M solution
R = 280 Ω
σ = ?
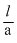 = 50 × 1.4 × 10-2
= 50 × 1.4 × 10-2
⇒ R = ρ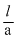
⇒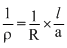
⇒ ρ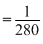 × 50 × 1.4 × 10-2
× 50 × 1.4 × 10-2
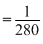 × 70 × 10-2
× 70 × 10-2
= 2.5 × 10-3 S cm-1
Now, λm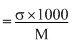
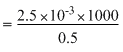
= 5 S cm2 mol-1
= 5 × 10-4 S m2 mol-1
R = 50 Ω
σ = 1.4 Sm-1 = 1.4 × 10-2 S cm-1]
⇒
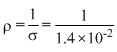 Ω cm
Ω cm Now, R = ρ
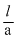
⇒
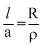 = 50 × 1.4 × 10-2
= 50 × 1.4 × 10-2 For 0.5 M solution
R = 280 Ω
σ = ?
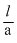 = 50 × 1.4 × 10-2
= 50 × 1.4 × 10-2 ⇒ R = ρ
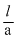
⇒
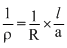
⇒ ρ
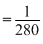 × 50 × 1.4 × 10-2
× 50 × 1.4 × 10-2 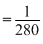 × 70 × 10-2
× 70 × 10-2= 2.5 × 10-3 S cm-1
Now, λm
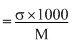
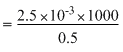
= 5 S cm2 mol-1
= 5 × 10-4 S m2 mol-1
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The correct cell diagram for the following reaction and Eo for the cell is2AgBr(s) + H2(g) → 2Ag(s) + 2H+ + 2Br−; $E_{Ag...How many gm of silver will be displaced from a solution of AgNO3 by 4 gm of magnesium?...A volume of 100 ml of 0.6 N−CuSO4 solution is electrolysed between two platinum electrodes till the concentration in the...When 0.1 mol MnO42- is oxidised the quantity of electricity required to completely oxidise MnO42- to MnO4- is...Element A (atomic mass = 112) and element B (atomic mass = 27) form chlorides. Solutions of these chlorides are electrol...