Gaseous StateHard
Question
Helium atom is two times heavier than a hydrogen molecule. At 298 K, the average kinetic energy of a helium atom is :
Options
A.Two times that of a hydrogen molecule
B.same as that of a hydrogen molecule
C.four times that of a hydrogen molecule
D.half that of a hydrogen molecule
Solution
Average k. E. = 3/2 KT and it depends on temperature only and not on the nature of gas. So both gas have same 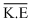 .
.
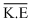 .
.Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Under which of the following conditions a real gas resembles an ideal gas -...The ratio between the r.m.s. velocity of H2 at 50 K and that of O2 at 800 K is:...If equal weights of oxygen and nitrogen are placed in separate containers of equal volume at the same temperature, which...Which nobal gas has maximum solubility in water :-...If P is pressure and ρ is density of a gas, then P and ρ are related as...