JEE Main | 2014Atomic StructureHard
Question
The correct set of four quantum numbers for the valence electrons of rubidium atom (Z = 37) is
Options
A.5, 0, 0, + 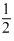
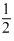
B.5, 1, 0, + 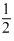
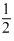
C.5, 1, 1, + 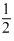
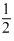
D.5, 0, 1, + 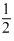
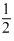
Solution
37 → 1s22s22p63s23p63d104s24p65s1
So last electron enters 5s orbital
Hence n = 5, l = 0, m1 = 0, ms = ±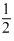
So last electron enters 5s orbital
Hence n = 5, l = 0, m1 = 0, ms = ±
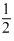
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Cathode rays are made up of electrons. Anode rays are made up of...If the shortest wave length of Lyman series of H atom is x, then the wave length of the first line of Balmer series of H...Deuterium nucleus contains :...An antiproton has the mass of a proton but a charge of –e. If a proton and an antiproton orbited each other, then how fa...In which transition, one quantum of energy is emitted ?...