Photoelectric EffectHard
Question
for maximum λ = 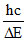
(n1 = 2 → n2 = 1 Lymen
(n1 = 3 → n2 = 2 Balmer
Longest λLymen =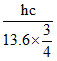
Longest λBalmer =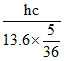 The electron in a hydrogen atom makes a transition from an excited state to the ground state. Which of the following statements is true ?
The electron in a hydrogen atom makes a transition from an excited state to the ground state. Which of the following statements is true ?
(n1 = 2 → n2 = 1 Lymen
(n1 = 3 → n2 = 2 Balmer
Longest λLymen =
Longest λBalmer =
Options
A.Its kinetic energy increases and its potential and total energies decreases
B.Its kinetic energy decreases, potential energy increases and its total energy remains the same
C.Its kinetic and total energies decrease and its potential energy increases
D.Its kinetic, potential and total energies decrease
Solution
Kinetic energy (K.E.) = 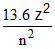
Potential (P.E.) =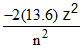
Total energy (T.E.) =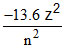
When an electron in H-atom makes a transition from an excited state to the ground state, value of n decrease, hence K.E. increases and its P.E. & T.E. decreases.
Potential (P.E.) =
Total energy (T.E.) =
When an electron in H-atom makes a transition from an excited state to the ground state, value of n decrease, hence K.E. increases and its P.E. & T.E. decreases.
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The radiation corresponding to 3 → 2 transition of hydrogen atoms falls on a metal surface to produce photoelectro...Chemical to electricalWhich one of the following is true in photoelectric emission :-...The orbital speed of the electron in the ground state of hydrogen is v. What will be its orbital speed when it is excite...A cesium photo cell, with a steady potential difference of 60 volt across it, is illuminated by a small bright light pla...Two photons having...